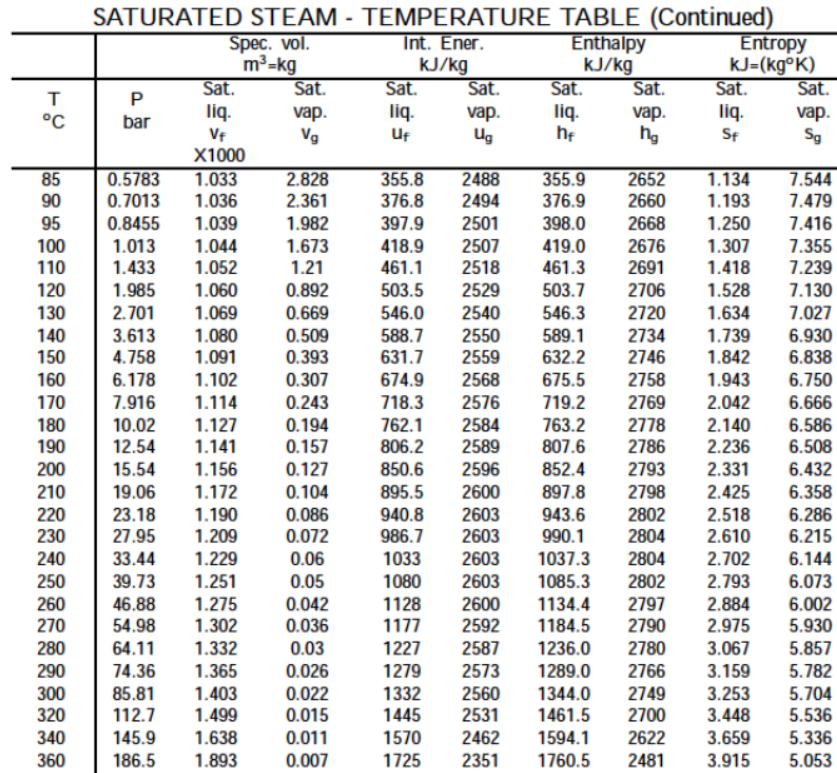 
A typical phase diagram for a pure substance is shown in Figure 1. These diagrams indicate the physical states that exist under specific conditions of pressure and temperature, and also provide the pressure dependence of the phase-transition temperatures (melting points, sublimation points, boiling points). A phase diagramcombines plots of pressure versus temperature for the liquid-gas, solid-liquid, and solid-gas phase-transition equilibria of a substance. Making such measurements over a wide range of pressures yields data that may be presented graphically as a phase diagram. Also described was the use of heating and cooling curves to determine a substance’s melting (or freezing) point. Considering the definition of boiling point, plots of vapor pressure versus temperature represent how the boiling point of the liquid varies with pressure. In the previous module, the variation of a liquid’s equilibrium vapor pressure with temperature was described. 
The physical state of a substance and its phase-transition temperatures are represented graphically in a phase diagram.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
